Antigen-Specific T Cells to Prostate-Specific Membrane Antigen (PSMA)
Our scientists were among the first to develop custom antigen-specific T cells for research use and have created them against a diverse range of targets, including viral, tumor, and self-antigens.
We’re now developing the next generation of antigen-specific T cells to target some of today’s most prevalent and challenging diseases. More on that below. But first, a primer on antigen-specific T cells.
The Role of Antigen-Specific T Cells in Disease Research and Treatment
T cells are a major player in the immune response in various diseases, including cancer and pathogenic infections, and are mediators in inflammation and autoimmunity. A large body of research has been conducted over the past few decades to manipulate T cell responses, leading to many clinical strategies including cancer vaccines, T cell adoptive therapy, CAR-T therapy, immune-oncology, and the use of regulatory T cells for autoimmune diseases.
Antigen-specific T cells are an important tool in T cell biology research and potency assays. Investigators can isolate them directly from the disease site (e.g., tumor-infiltrating lymphocytes (TIL) or inflammatory lesion infiltrate populations), but these procedures often yield a limited number of cells due to low cell frequency and lack of robust isolation methods.
Alternatively, antigen-specific T cells can be generated in vitro using antigen-presenting cells pulsed with whole antigens or peptides. However, this process is often time-consuming with a low success rate.
About Ignyte Bio’s Antigen-Specific T Cells
Ignyte Bio offers antigen-specific T cells for research use you can rely on.
- Typically generated using multiple in vitro stimulations with peptide antigens
- Not immortalized or genetically modified, so they more closely mimic physiological T cells
- Specificity analyzed using the cognate peptide/MHC tetramer binding as well as interferon-gamma (IFN-γ) secretion assays.
Prostate specific membrane antigen (PSMA) is a 750 amino-acid, type II glycoprotein highly expressed in prostate adenocarcinoma cells. It is associated with high grade, metastatic prostate cancer. PSMA has emerged as a useful target in PET imaging of prostate cancer especially in the evaluation of small volume lymph node and bone metastases using tracers like Ga-68 PSMA and F-18 PSMA. The theranostic (combination of therapeutic and diagnostic) agent Lutetium-177 vipivotide tetraxetan (trade name Pluvicto) used to treat metastatic castration-resistant prostate cancer obtained FDA approval in 2022.
For the interest of identifying T cell epitopes destined to become immunotherapeutic for prostate cancer, computer-based algorithms have been used to predict possible PSMA peptides capable of stimulating specific CD8 T Cells restricted by HLA-A2. To date, several identified PSMA peptides have the capacity to induce a cytolytic T lymphocyte (CTL) response in vitro. However, only one peptide (PSMA27-35 : VLAGGFFLL) can induce specific CD8 T cells capable of recognizing prostate tumor cells that express PSMA and HLA-A2. One group reported a clinical trial involving a DNA vaccine encoding a domain (DOM) from fragment C of tetanus toxin linked to PSMA27-35 (Chudley et. al., 2012).
Ignyte Bio offers the Antigen-Specific CD8 T Cells (ASpecT) against the PSMA27-35 : (VLAGGFFLL) peptide, restricted by HLA-A2 protein.
Figure 1 shows specific recognition of the PSMA27-35 (VLAGGFFLL) in association with HLA-A-02:01 as shown by the tetramer binding study. Figure 2 shows specific IFN-γ secretion and specific lysis when HLA-A-02:01+ target cells were incubated with the PSMA27-35 peptide but not the control MART-126-35, 27L (ELAGIGILTV) peptide.
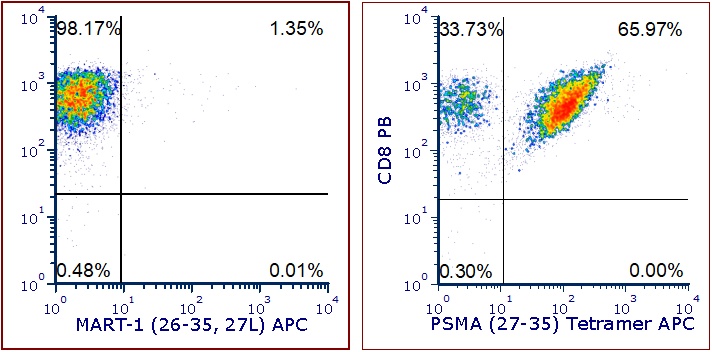
Figure 1. Recognition of Antigenic Peptide PSMA27-35 Demonstrated by Peptide/MHC Tetramer Binding Analysis. Cryopreserved T cells were thawed and stained with APC-labelled HLA-A-02:01-PSMA27-35 (VLAGGFFLL): PSMA (27-35) Tetramer APC or a control tetramer HLA-A-02:01-MART-126-35, 27L (ELAGIGILTV): MART-1 (26-35, 27L Tetramer APC). These cells were counterstained with anti-CD8 Pacific Blue reagent and analyzed by flow cytometry. Non-viable cells were excluded from the analysis using 7-AAD.
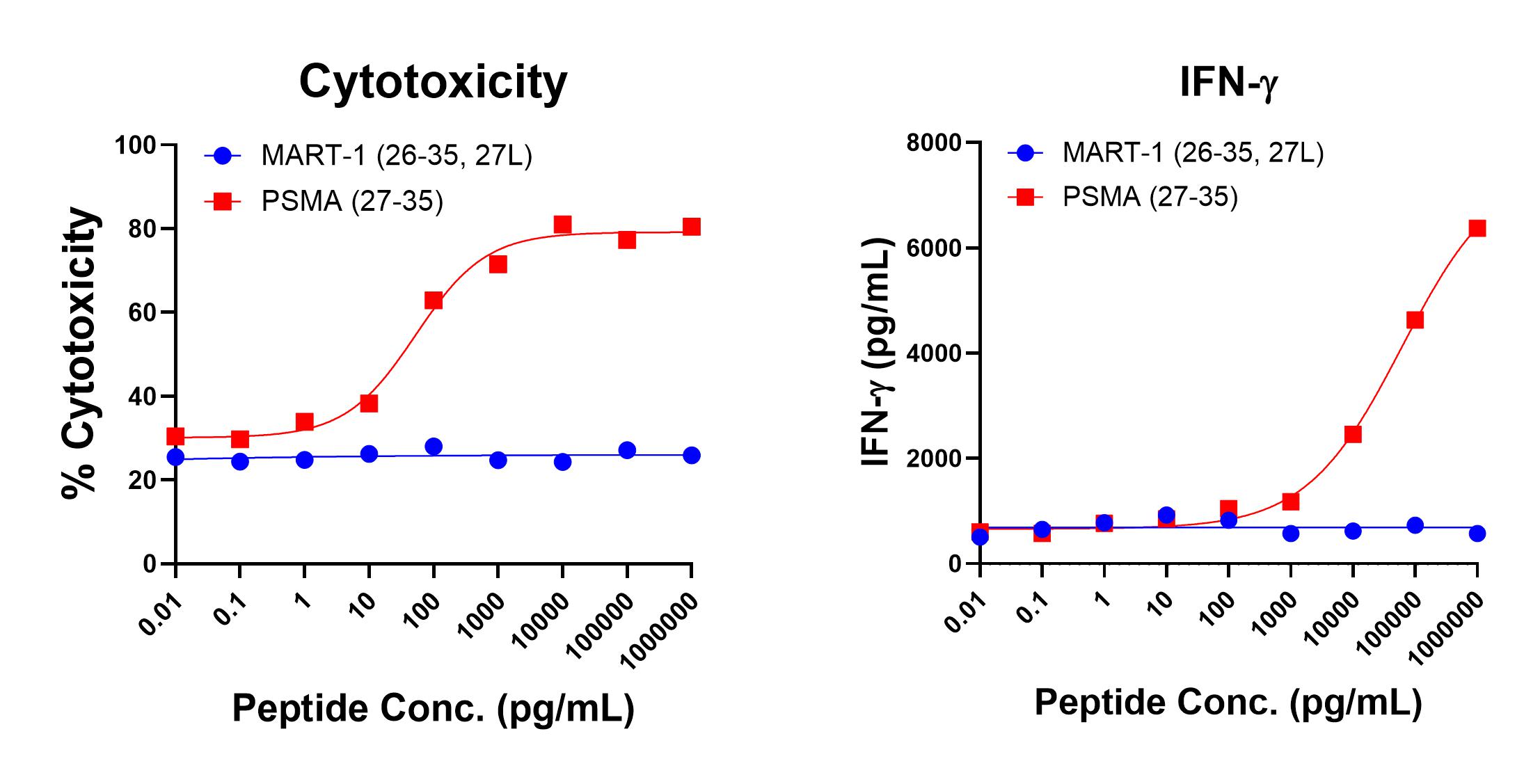
Figure 2. Antigen-Specific IFN-g Secretion and Cytotoxicity Data. Freshly thawed anti-PSMA27-35 T cells were plated at 2 x 104 cells/well in the presence of 2 x 104 T2 cells, an HLA-A-02:01+ B-LCL (ATCC, Manassas, VA) and different concentrations of PSMA27-35(VLAGGFFLL): PSMA peptide or MART-126-35, 27L (ELAGIGILTV): MART-1 control peptide in a 96-well round bottom plate. After 18-24 hours of incubation a t 37oC, 5% CO2, culture supernatants were harvested and assayed for IFN-g using the Lumit™ IFN-γ Immunoassay (Promega, Madison, WI). IFN-γ concentration is plotted against CEA and control peptide concentrations (right graph). Remaining cells in each well were stained with the viability dye 7-AAD and analyzed using flow cytometry. % Cytotoxicity (= % 7-AAD+ target cells) is plotted against the respective peptide concentrations (left graph).
