Antigen-Specific T Cells for Melanoma Research: Tyrosinase
Our scientists were among the first to develop custom antigen-specific T cells for research use and have created them against a diverse range of targets, including viral, tumor, and self-antigens.
We’re now developing the next generation of antigen-specific T cells to target some of today’s most prevalent and challenging diseases. More on that below. But first, a primer on antigen-specific T cells.
The Role of Antigen-Specific T Cells in Disease Research and Treatment
T cells are a major player in the immune response in various diseases, including cancer and pathogenic infections, and are mediators in inflammation and autoimmunity. A large body of research has been conducted over the past few decades to manipulate T cell responses, leading to many clinical strategies including cancer vaccines, T cell adoptive therapy, CAR-T therapy, immune-oncology, and the use of regulatory T cells for autoimmune diseases.
Antigen-specific T cells are an important tool in T cell biology research and potency assays. Investigators can isolate them directly from the disease site (e.g., tumor-infiltrating lymphocytes (TIL) or inflammatory lesion infiltrate populations), but these procedures often yield a limited number of cells due to low cell frequency and lack of robust isolation methods.
Alternatively, antigen-specific T cells can be generated in vitro using antigen-presenting cells pulsed with whole antigens or peptides. However, this process is often time-consuming with a low success rate.
About Ignyte Bio’s Antigen-Specific T Cells
Ignyte Bio offers antigen-specific T cells for research use you can rely on.
- Typically generated using multiple in vitro stimulations with peptide antigens
- Not immortalized or genetically modified, so they more closely mimic physiological T cells
- Specificity analyzed using the cognate peptide/MHC tetramer binding as well as interferon-gamma (IFN-γ) secretion assays.
Announcing Ignyte Bio’s Newest Antigen-Specific T Cell Line for Melanoma Research: anti-Tyrosinase CD8+ T Cells
Melanoma is one of the malignancies that have been studied intensively, particularly in terms of the immune response to tumors, and has been used as a model for the development of immunotherapy.
Read more about our
Tyrosinase specific CD8+ T cells available for purchase.
About the Tyrosinase Antigen
Tyrosinase is copper containing enzyme expressed in melanocytes, responsible for the first step in melanin production. It converts the amino acid tyrosine to dopaquinone. A series of additional chemical reactions convert dopaquinone to melanin in the skin, hair follicles, iris, and the retina. In addition to melanocytes, tyrosinase is expressed in nearly all fresh melanoma tumor samples as well as in 70% of melanoma cell lines. The tyrosinase (TYR) gene encodes antigens recognized by anti-melanoma cytolytic T lymphocyte (CTL) clones as initially reported by Brichard et. al., 1993.
One of the well-studied tyrosinase CTL epitopes is a peptide that represents residues 368-376. The naturally occurring HLA-A*0201-associated peptide was identified by mass spectrometry in eluates of melanoma cells. The sequence of the eluted peptide differed from the gene-deduced sequence at residue 370 (aspartic acid instead of asparagine. 370D) as a result of a posttranslational modification. It was also reported that recognition of the natural tyrosinase peptide YMDGTMSQV by a specific CTL clone derived from an autologous mixed lymphocyte-tumor cell culture was ~100-fold more efficient than that of the gene-deduced peptide YMNGTMSQV (Skipper et. al., 1996).
Ignyte Bio’s anti-Tyrosinase-specific CD8+ T cells are generated against the natural tyrosinase peptide TYR368-376, 370D, (YMDGTMSQV). Note: in some publications, this peptide is also known as tyrosinase 369-377, 371D.
Figure 1 shows specific recognition of the TYR368-376, 370D in association with HLA-A-02:01 as shown by the tetramer binding study. Figure 2 shows specific IFN-γ secretion and specific lysis when HLA-A-02:01+ target cells were incubated with the TYR368-376, 370D peptide but no the control MART-126-35 ELAGIGILTV peptide.
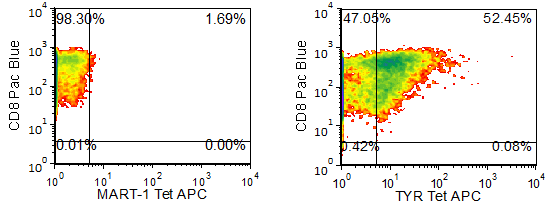
Figure 1. Recognition of Antigenic Peptide (Tyrosinase368-376, 370D) Demonstrated by Peptide/MHC Tetramer Binding Analysis. Cryopreserved T cells were thawed and stained with the tetramer HLA-A-02:01-Tyrosinase368-376, 370D (YMDGTMSQV, TYR Tetramer APC or the APC-labelled HLA-A-02:01-MART-126-35, 27L(ELAGIGILTV, MART-1 control Tetramer. These cells were counterstained with anti-CD8 Pacific Blue reagent and analyzed by flow cytometry. Non-viable cells were excluded from the analysis using 7-AAD.
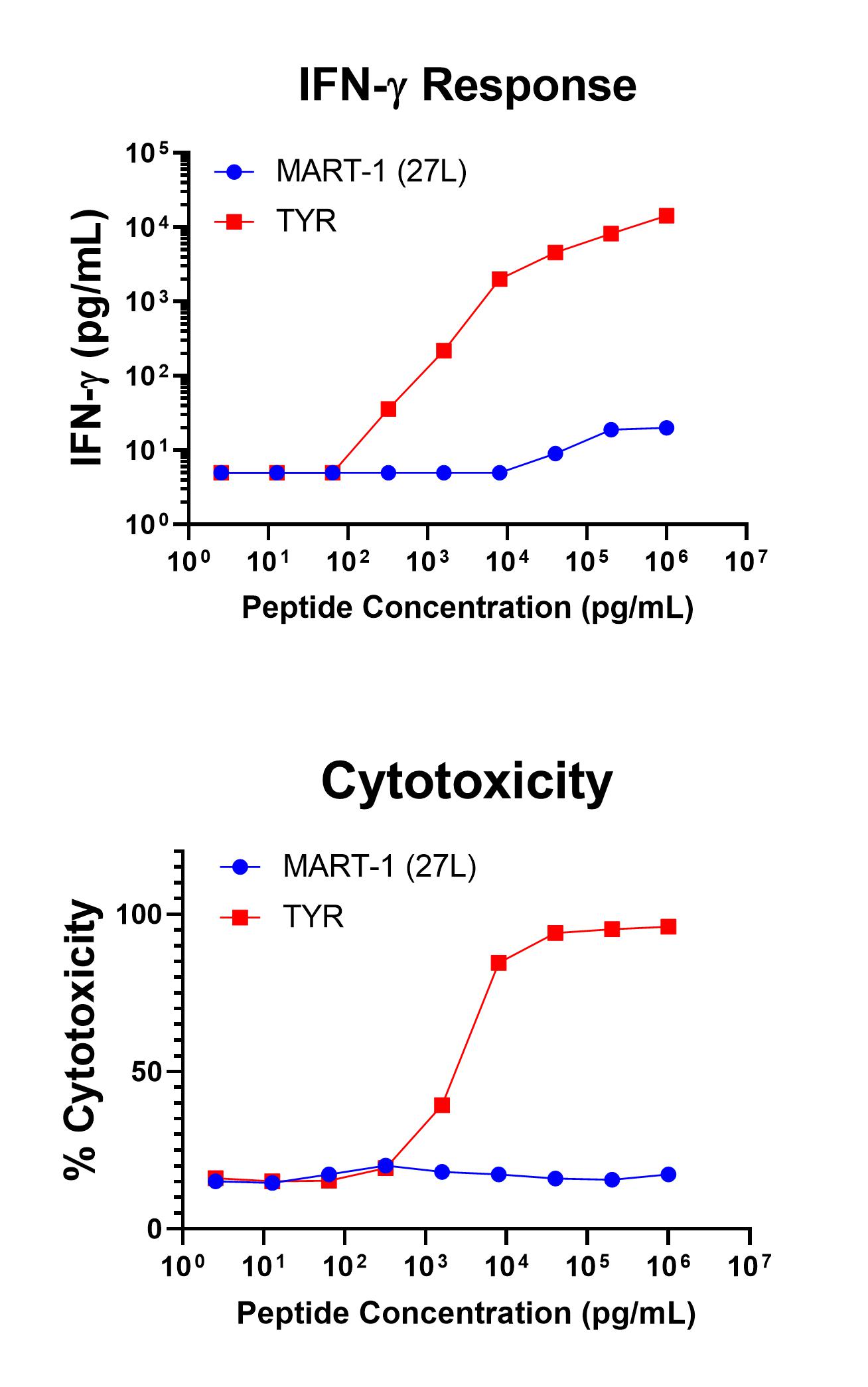
Figure 2. Antigen-Specific IFN-γ Secretion and Cytotoxicity Data. Freshly thawed anti-Tyrosinase CD8+ T cells were plated at 2 x 104 cells/well in the presence of 2 x 104 T2 cells, an HLA-A-02:01+ B-LCL (ATCC, Manassas, VA) and different concentrations of Tyrosinase368-376, 370D (YMDGQMSQV): TYR or MART-126-35, 27L (ELAGIGILTV): MART-1 (27L) control peptide in a 96-well round bottom plate. After 18-24 hours of incubation at 37oC, 5% CO2, culture supernatants were harvested and assayed for IFN-γ using the Lumit™ IFN-γ Immunoassay (Promega, Madison, WI). IFN-γ concentration is plotted against Tyrosinase and control peptide concentrations (upper graph). Remaining cells in each well were stained with the viability dye 7-AAD and analyzed using flow cytometry. % Cytotoxicity (= % 7-AAD+ target cells) is plotted against the respective peptide concentrations (lower graph).
